Ready...
Set...
EvidenceTM
Outcome synthesis for teams working with real-world evidence, including biotech, medical device, pharmaceutical, and any group that needs to organize endpoints, compare cohorts, and turn messy evidence into decision-ready outputs.
Endpoint Organization
Add endpoints from multiple studies into one structured workspace, align related evidence across sources, and make it easier to compare cohorts, subcohorts, and outcome patterns without losing the underlying context.
Outcome Synthesis
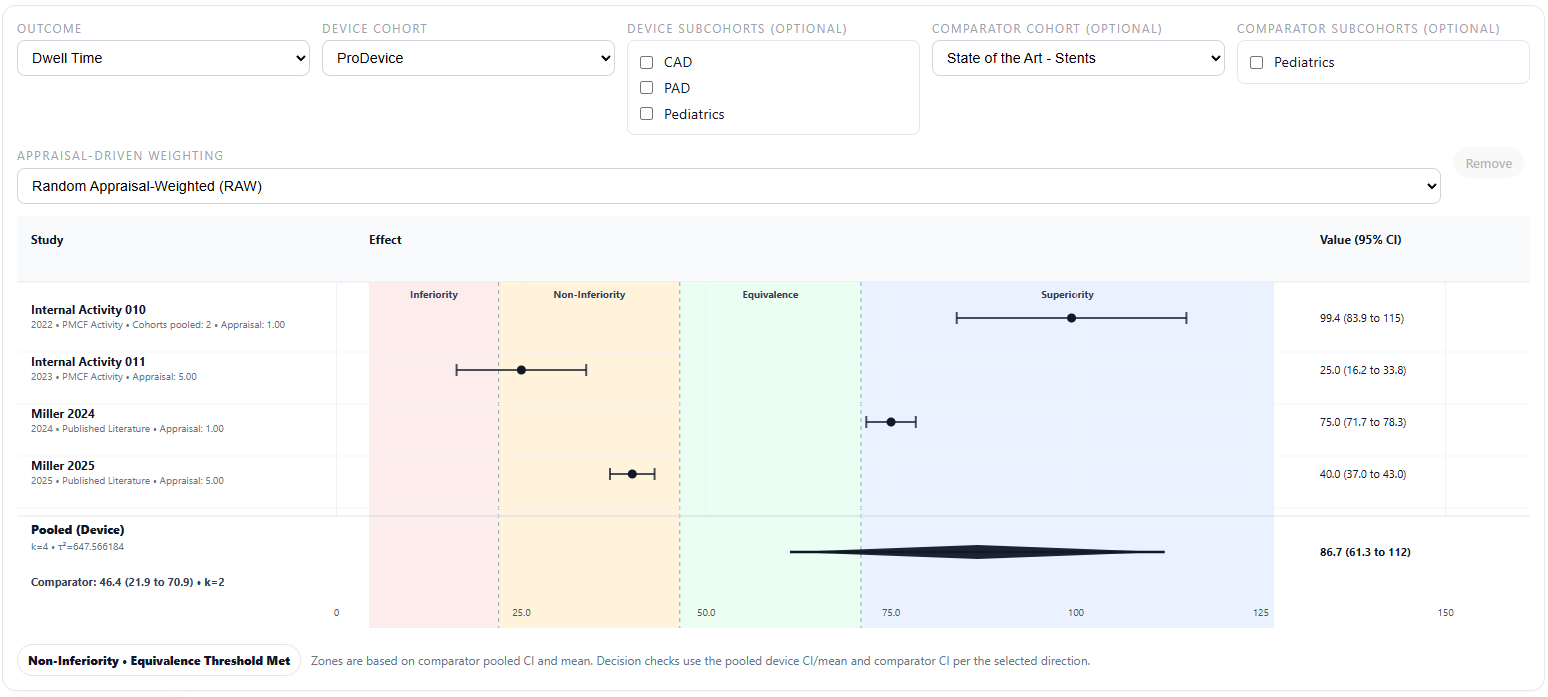
Ready...Set...EvidenceTM Outcome Synthesis is not limited to clean controlled studies. It is built to synthesize uncontrolled data as well, reflecting the kinds of outputs that real-world evidence often produces. It also incorporates the Random Appraisal-Weighted (RAW) Method1.
Browser-Based Workflow
The synthesis module, and future modules, are designed to let regulatory, clinical, and post-market surveillance teams work directly in the browser through a practical interface built for evidence handling rather than generic document storage.
Outcome Synthesis Built for Real Evidence
Compare control and comparator cohorts, explore subcohorts, synthesize uncontrolled and published data, and surface patterns that are easy to miss when evidence is scattered across studies and endpoints. The goal is not just to store data, but to make meaningful comparison and synthesis faster and more transparent.
The Beta Synthesis Module Is Open Access
The beta synthesis module is currently open access. Beta access to additional modules is available upon request.
To get started, create an account with an email address and password.
Available Now
- Open-access beta use of the synthesis module
- Endpoint organization across multiple studies
- Cohort and subcohort comparisons
- Outcome synthesis for real-world and uncontrolled data
- RAW Method integration1
What’s Coming / Paid Access
- Team management and multiple-user environments
- Advanced user permissions and administrative controls
- Shared workspaces and collaboration tools
- Enhanced data backup and recovery options
- Higher uptime environments and priority infrastructure
- Expanded storage and long-term retention options
- Clinical study management
- Evidence coverage
- Registrations
- Regulatory flow charts
- Complaint management
- Feedback analysis
